-
Mastering Fire Safety: Preventing and Reacting to Fires Promptly
Fires can occur at any time and in any place, making it crucial for individuals and organizations to be well-prepared for these unpredictable events. By understanding the key principles of fire prevention and learning how to react to fires quickly, significant damages and injuries can be minimized or even prevented altogether. Comprehensive Strategies for Fire Prevention Preventing fires before they start is the best way to protect your property and occupants from harm. Here are some essential elements of a successful fire prevention strategy: Regular Inspection and Maintenance of Equipment Keeping equipment in good working order reduces the risk of fires starting from mechanical failures. Make sure that all machines,…
-
Do You have What it takes to become a Winemaker?
Many people at some point in their life fall in love with the idea of owning a vineyard and becoming a winemaker. However, it is a long process that usually involves many different elements to take place, namely an education in the winemaking process, some experience in the field (literally) and a passion for the craft. Without the latter one, it is more than probable that a person won’t last in this industry, since it requires hours and hours of work. Here is what you will need if you want to become a winemaker. An Education in Winemaking Every profession requires an education. Of course, if you are born in…
-
What are the Benefits of Electronic Manufacturing Services?
When a company wants to create a product that features electronic components inside, they often call upon the services of an electronic manufacturing service provider. With the experience and the knowledge that they have accumulated in the field, they can help them create better products and make sure that they meet all necessary standards in the industry. Here are some of the benefits that EMS offer to their clients. Reduced Costs Designing and producing a product including electronic components can be really expensive. First, you need to make sure that the design of the product will not only be optimal for the end user, but also functional. This cannot be…
-
Why Americans from California love travelling to France?
Sometimes, people travel because they want to find themselves in a completely different environment than the one they evolve in daily. But other times, they head to a destination that they have ties with. That is the case with people from California who love to visit France, because of the elements that bide them together, namely culture, wine and technology. Let’s take a closer look at the reasons why US West Coasters fly across the Atlantic Ocean to visit France. Culture California is a land of culture; so is France. Hollywood might be the center of the world when it comes to cinema, but everyone knows that French directors are…
-
Unlocking the Power of Female Entrepreneurs: France, Germany and the UK
In recent years, there has been a steady increase in female entrepreneurs making their mark on the European business landscape. As more women take the leap into entrepreneurship, it’s essential to explore the environment that fosters their success. In this article, we’ll delve into the business climates in France, Germany, and the UK to determine where women are most successful in business. The Rise of Women in Business According to Reboot Online, a digital marketing agency, there has been a significant shift in the number of women starting and successfully running businesses in Europe. This trend is expected to continue as more support systems and resources become available for female…
-
Girls and Airsoft: Breaking the Gender Barrier in an Exciting Sport
In recent years, airsoft has gained popularity as a thrilling sport that combines teamwork, strategy, and physical fitness. While it is often perceived as a male-dominated activity, more and more girls and women are discovering the excitement of airsoft and challenging gender stereotypes. This article will explore how female players are making their mark in the world of airsoft and why this sport is indeed suitable for both men and women. Airsoft: An Inclusive Sport for All Genders The key to understanding why airsoft is a suitable sport for everyone lies in its nature. Unlike some sports that heavily rely on physical strength or speed, airsoft focuses on skills such…
-
How to create a Successful e-boutique
You have finally decided that you want to have your own business and an e-boutique sounds like the right solution for you. There is no doubt that many opportunities are still available online, but if you want to succeed, you will have to put all the odds in your favour by creating a website that is efficient for your future customers. Here is how to build a successful e-boutique. Insert a Chatbot If you want your website to compete with other companies in the same sector, you need to make customer service a priority. In the technological world that we live in, it starts by inserting a chatbot where they…
-
Why You should try ChatGPT
It is almost impossible to not have heard about ChatGPT by now. It is one of the hottest topics of 2023, and it should remain in the news for years to come, as the AI keeps learning and growing. It interests everyone from Elon Musk to the EU legislators who are looking to create the first rules and regulations in regards to it. In the meantime, if you haven’t tried it yet, here is why you should. Your Friend ChatGPT As its name mentions it, ChatGPT is a communication tool that can answer any questions that you may have. Many people have already adopted it as something that they can…
-
What You need to know if You want to study in Germany
No matter what your age is, you may be thinking of going to study in Germany to complete your degree and to enjoy a new life experience at the same time. There are many reasons that can justify that choice. Here is the information that you need, if you want to make that life-changing decision. Cost of living Although Germany is the economic center of Europe, it is not the most expensive place to live. Naturally, the cost of living will vary on the city that you choose. Berlin is known to be one of the most affordable cities to live in, for students. You should be able to stay…
-
Trending Watches in 2023
Everything has changed after the pandemic – it is just a simple fact. It doesn’t mean that fashion is not trending backwards though, at least in some regards. You can definitely see the dichotomy in the world of watches, as sustainability and gender-neutral models are two words that are modifying watches style, while retro glamour is also popping back up on the scene. Here are the trendy watches of 2023. Welcome to the Modern World If you have not heard anything about those AI noise canceling call watch yet, let us introduce them to you. As you know AI is infiltrating itself everywhere these days. Just in the first few…
-
Benefits of Playing Soccer to the Youth
Each year, millions of children in the United States participate in youth soccer. According to recent data, it is the most popular youth league sport in the country. Why? Since soccer is among the most widely played sports in the world and also most kids are excited to wear and have their own psg jersey. This is an excellent approach to introducing children to team sports. Even if kids do not pursue soccer for an extended period of time, the advantages of playing for teams such as FC Highland may remain for years. These are nine advantages of young soccer participation: The Benefits Soccer Offers Cardiovascular endurance There are several…
-
Best Outfits for Plus Sizes to Wear with Thigh High Boots
In a footwear-obsessed culture, boots are a must-have wardrobe since they complement every ensemble, regardless of the occasion or event. The nicest feature is that these sneakers may be worn in practically any season! You may use them to effortlessly create a professional or even a casual look. Yet, we have observed that while females are comfortable carrying boots, they are terrified by thigh-high boots. It might be difficult for curvaceous women to locate plus size thigh high knee high boots outfit that complement them. If you’re unsure of how to match your thigh-high boots with various clothes, we’ve compiled a list of the greatest outfits that you may follow…
-
Great Gifts for All Ages of Anime Lovers
If you’re searching for a unique present for an anime enthusiast, you may be at a loss for ideas. This is especially true if you’re new to the realm of Japanese animation or if you don’t know much about animation. Fortunately, we’re here to present the greatest anime-themed gifts on the market, making your chore a little bit easier. Whether the recipient is a devoted fan of Naruto, Dragon Ball, One Piece, or My Hero Academia, you will discover a present that they will adore. Our selection includes a variety of items, from charming accessories and clothing like anime shorts or robes to amusing décor. We are aware that there…
-
What are forex breakouts?
In forex trading, breakouts are a critical part of your success. But what are they, and how do you take advantage of them? In this article, we’ll explain everything you need to know about breakouts so you can start trading like a pro. Keep reading to find out.
-
Luxury Italian Fashion Brand FENDI Keeps Landscape Preservation Close to Heart
As a brand deeply rooted in the Italian territory, FENDI has the preservation of the beautiful Italian landscapes at heart, in just one example, requalifying an abandoned industrial site in the hills of Tuscany to build its “new pole of excellence”—a future production site—distinguished by state-of-the-art environmental performance.
-
Runway Walk – How To Walk Like Iconic Supermodels?
To walk the walk on a runway show is not as easy as one might think. The shoes are almost always very high heels, and you need to be in perfect shape and have a confident posture. As a matter of fact, it can take hours of practice before being able to walk on a runaway of a big fashion show. However, if you want to try it out for yourself, don’t worry, there are some tricks to get a least a good basic understanding of how to walk the walk like a professional pageant. Continue below to know more.
-
Complete Guide: How to Do a French Manicure
It is true that freshly done nail polish can go a long way in making a good impression on others. It is the kind of small detail that we don’t always think about at first, but we do notice beautiful nails from time to time. All that said, a big question remains: is a French manicure really French? Find out everything you need to know about nail painting and the history of French manicures in the following guide.
-
Top 10 Best Vampires Movies on Netflix 2021
Have you ever wondered when were vampires invented? These blood-sucking creatures go back a very long way, as far as the 17th century, actually! Tales of Dracula are known in the entire world and have even inspired the famous video game series of Castlevania. If you’re looking for a good vampire flick to watch on Netflix, we got your back! Here’s our top 10 list of the best Netflix vampire shows and movies.
-
Top 10 Wish Alternatives for Shopping
We all love spending money, and what better way to do that than an application where everything seems cheaper than usual? Wish is a very popular selling platform, but let’s be honest, what’s on the picture and the product that you get at home are often quite different, to say the least. That said, there are many other shopping apps you can use that are as good, or even better than Wish. Here are our Top 10 Apps like Wish that we love using.
-
What is the Bohemian Lifestyle?
Being a bohemian does not mean you dress like one. It is something that has to do with lifestyle and how you think. You might consider adopting this lifestyle or wonder if the way you live your life is considered Bohemian. Keep reading to know more about the bohemians.
-
How to make distilled water at home?
It is not uncommon to hear that water is important for your body and that you should always drink enough water every day to stay hydrated. That said, apart from the tap and bottled water, you can also drink distilled water. Of course, the latter is absolutely safe to drink, yet, it lacks the minerals you need because of the distillation process. Moreover, it has no flavor and it’s pretty flat. However, if you don’t mind the taste, know that you can make your own distilled water at home, and that’s what we are gonna talk about down below!
-
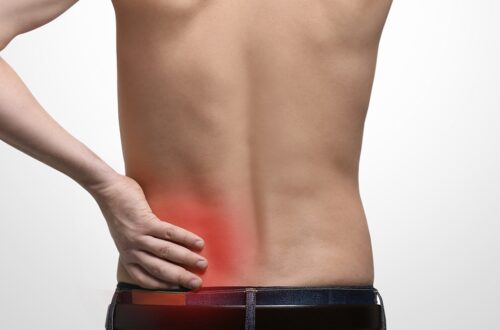
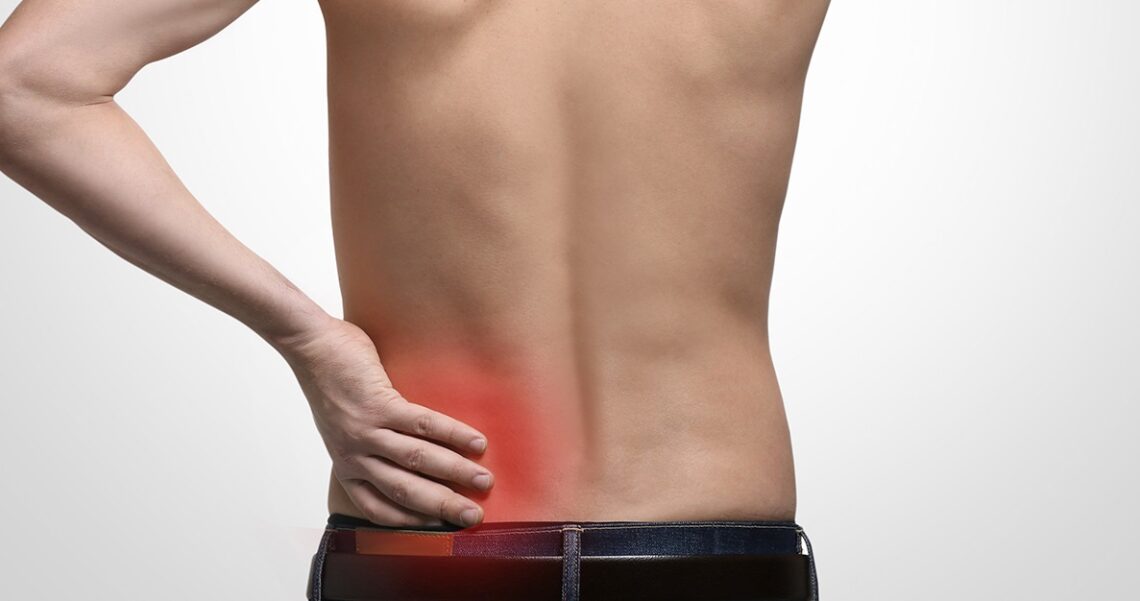
How to ease Degenerative Disc Disease Pain Naturally
Degenerative disc disease or disc desiccation is a symptom of lower back and neck pain. This occurs when your spinal disc tears and wears from dehydration as the vertebral discs should be full of fluid to keep them flexible and strong. Losing the discs’ fluid makes your back feel stiff, weak, tingling, numb, and especially painful. Of course, there are many causes behind this disease. If you were wondering if there are any solutions to ease the pain, continue reading because we are going to show you 3 easy ways to naturally alleviate disc disease pain.
-
What does encountering a Cardinal bird Symbolize?
You might have heard of this beautiful bird species that is the cardinal, and considering everything revolving around the famous animal, it is no surprise. Majorly located in Northern America, the cardinal presents a distinctive color feature, making it easy to spot from afar. It is certainly one of the most recognizable birds in the region with its black mask coupled with a bright red feather coat. From the many myths and superstitions involving this beautiful bird, we created this guide that we hope will answer most of your questions about this creature.
-
Can being in a couple translate into fulfilment?
Many studies have been done over the last decade, regarding how being in a couple affects individuals. Most were searching to understand if their psychological and physical health was better or worse. It would seem that the answer is: People fare better, generally speaking, when they share their lives with a significant other. Here are the various ways it helps each individual reach fulfillment.
-
The best secure websites to watch anime
As you may already know, there are tons of free anime websites available on the internet. However, you might be worried that these websites contain spyware or viruses that could harm your computer. No worries, there are safe streaming services available on the web, and we will share them with you in our top 10 down below. Let’s start!
-
Home Depot Engine Crane Services
Home Depot is the biggest home improvement distributor in the United States, providing tools, construction products, and various services. Moreover, Home Depot also offers an engine crane service for people who would like to rent one. With the help of such equipment, you could be installing, removing, or lifting your car’s engine in no-time! The engine crane can also be used with heavy items such as boxes and woodworks. Using a big machine like that not only helps save your time but also helps reduce hazards. In this article, we will inform you about the different types of engine cranes, how to rent and buy an engine crane from Home…
-
Tool for rent: Lowe’s Rental Equipment Program
Many people may face a problematic situation when something is broken in the house. In general, it is due to the fact that they have the tools required for the reparation, or they know how to fix it. On top of that, the tools are mostly expensive. One could wonder: Is it really worth it for just one job? Actually, know that you don’t have to spend a lot of money to get the tools that you need. Indeed, you can simply rent them from a tool store. That way, you won’t have to waste your money on one little repair job in the house! There are special Lowe’s stores…
-
How to do the most 3 popular vape tricks like a pro?
There are many vape smoking tricks you might have seen in movies, music videos, or from a vaper who was trying to impress you by showing off his skills. They make vapor smoke turn into magical shapes and videos of such performances became viral very quickly. Moreover, newer vaping devices can produce deeper vapor smoke, which is way more effective than cigarette smoke. It indeed offers a thicker cloud and it makes it easier to perform some neat tricks. In this article, we are going to show you the 3 most well-known vape tricks and how to pull them off. That way, you will become a vape master!
-
Everything you need to know about the 2017 Chevy Camaro Z28
The Chevrolet Camaro is an iconic pony also known as a mid-size muscle car. It is an American automobile manufactured by Chevrolet General Motors. The famous car first went on the market on September 29th, 1966. The 1967 model was designed for competing against the Ford Mustang of the same year. In 2012, Chevrolet finally unveiled its Camaro ZL1 for the first time. This reveal was later followed by another coup de grâce, the return of the Camaro Z/28. Although this robust machine had an honest success among fans of the series, it also had its share of doubts leading to its release. Let’s find out why!
-
Why are Crotch Rocket Motorcycles ending?
Some people love sportbikes as they look really cool on the road. Even though those who love speed have a strong desire to ride these sport-motorbikes, the end of the crotch rocket seems to be near. In the following article, we are going to inform you of the crotch rocket motorcycles; their prices, their history, as well as why it seems like it is the end of the road for them in the motorcycle industry. Let’s start!
-
Are the Great Courses Plus and the Great Courses good for learners?
People around the world have to stay home as the global pandemic persists, and it seems that we still have a long way to go despite the availability of vaccines. While it is a very odd situation for many, it is also an opportunity to learn something new like a new language or a new recipe, or learning how to play an instrument, and so on. If you are stuck at home because of COVID-19, perhaps it is the best time for you to learn a new skill or a new passion. That said, you may wonder what e-learning website is best for you? In fact, the 2 most popular…
-
How To Impress Your Crush with Jokes
Everyone loves people with a sense of humor because they can make you laugh and happy which is good for the heart. That said, wouldn’t it be great to have some funny jokes to tell your crush or your girlfriend/boyfriend? Moreover, it could help your conversation flow better as well. In this article, we will provide you multiple lists of funny pick-up lines, corny jokes, and great puns that you can try with anyone you like. Oh and don’t worry, these have been tested beforehand (not really).
-
The Vatican and Pope Francis: truths about their net worth
Every religion influences our beliefs. The belief varies from one individual to another. That said, in a majority of countries, we are mostly free of believing what we want, as long as it doesn’t harm anybody. The Vatican City, a city-state within Rome, houses some of the most beautiful pieces of art and architecture in the world. Besides its art and museums, it is best known as the home of the Pope and the Catholic Church. However, what is hiding behind the Roman Catholic Church headquarters? How wealthy and pious the Vatican and Pope Francis really are? Let’s find out in the following article.
-
Resolve error messages of Openload Stream Authorization
In the past, watching your favorite videos, movies, or TV series was perhaps a bit more complicated than nowadays. Streaming platforms are now used all around the world such as Disney Plus, Apple TV, Netflix, HBO Max, and Amazon Prime as they provide the content you want to watch. The only requirement: you need to pay a monthly subscription to get a membership. But what if you wanted to access all this content for free? That’s right, internet users who don’t want to pay to access said content can do so by using Openload Stream Authorization or an Openload co-pairing with the Kodi app. The latter is a very popular…
-
4 Solutions to find your student loan account number
Many people say that “Education is worth the financial investment” and that student loans are considered as good debts. Nowadays, there are more than 45 million borrowers with a total of $1.6 trillion students in debt in the United States. On average, statistics have found that it takes around 20-years for a former student to be free of debt. However, after graduation, parents and family are often affected by this situation and will help the student repay his/her loan. But, while you’re doing your best to repay all this, there might be a time when you will need some very specific info. Indeed, what if you were simply looking for…
-
Review Top Ranked PS4 Games for Girls
Gaming provides more benefits than one might think. It indeed helps relieve stress and boost our daily mood. Imagine you come back home from the office, and you have a big library of PS4 titles just waiting for you after a tough day at work. Sounds pretty good, doesn’t it? However, many people still misunderstand video games and may think that they are only for men or for kids. Nowadays, PS4 consoles (and video games in general) have become essential items in the living room. That said, if you are looking for something in particular, you found the right place! Down below, we listed for you our top 10 recommended…
-
Silmarillion, a new upcoming movie from JRR Tolkien’s books?
JRR Tolkien has brought us one of the most admirable worlds to date. “Middle-Earth” was partially depicted through The Lord of the Rings and The Hobbit trilogies. Turning books into film series since 2001, Peter Jackson has succeeded to present Tolkien’s universe to the masses. Indeed, these films are some of the most influential movies ever in history. Throughout Middle-Earth tales, we are introduced to the 3 world’s ages: the first age, the second age, and the third age (The Lord of the Ring and The Hobbit’s era), and the fourth age which begins after the defeat of Sauron. Although we already saw quite a bit through the 6 extended…
-
DIY: How to Decorate Your Futon Cover
A futon is originally from Japan. It is a type of mattress with multiple uses: it can be a mattress, a convertible bed, a platform bed, or even a sofa bed. Due to their useful and comfortable feathers, futons became a common necessary piece of furniture in many households around the world. Since futon covers come in a wide variety of styles, not only they protect your mattress, but they also serve as house decoration. Today, we gonna share with you some DIY ideas to make your own futon and redesign your room. Let’s begin, shall we? Interesting DIY Futon cover ideas Classic white sheet cover A white sheet cover…
-
5 Homemade Recipe Ideas with Rosehip
When we talk about roses, the queen of flowers, we usually think about their beauty, their pleasant fragrance, and of course, Valentine’s Day. In fact, the rose is one of the most ancient medicinal plants and has been used in many countries around the world like ancient Rome, Persia, India, China, and many more. Interestingly, many people don’t know that roses produce seeds like other flowers. Actually, these seeds are called rose hips/rosehips. The latter have useful medicinal properties, thus why they are widely used for producing medicines and cooking as well. Today, we reveal all the secrets behind the rosehips in the following article.
-
Estafiate: Miracle Herb & Its Healing Powers
Even with all the healthcare modern innovations, a lot of people nowadays tend to turn back to traditional herbal remedies. Instead of consuming manufactured medicines often containing chemical substances, people prefer natural herbs that generally offer a safer treatment. In fact, our ancestors have registered a long list of useful medicinal herbs. This indigenous knowledge has been transmitted from generation to generation. One of the most effective and therapeutic medicinal plants in the world bears the name “Estafiate”. Throughout this article, we will talk about everything this small herb can do.
-
How Can I Solve The Constant Sweet Taste In My Mouth?
Everybody loves sugary treats, we naturally enjoy the sweet taste in our mouths. However, some of us get a sweet taste without consuming any kind of sweets. This can be alarming regarding your health and is worth getting a look into. Therefore, we prepared this article to answer all your questions on this subject.
-
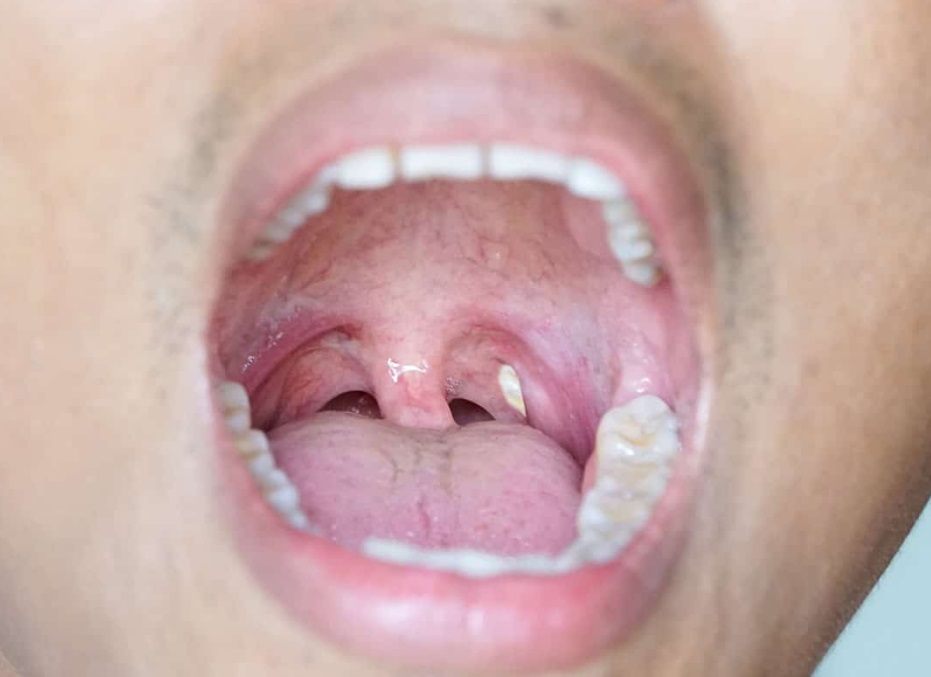
Easy Tips to heal from Tonsil Stones
Tonsil stones, also known as tonsilloliths, are off-white waste that look like pimples normally found in the back of the throat. If you are wondering what tonsils are exactly, they are the two little masses of tissues that help your immune system defend itself against bacteria and infections. Although tonsils stones are not dangerous, many people suffer from bad breath or from a sore throat because of them. You’re struggling to get rid of your tonsil stones? Don’t worry, we have the solution for you in this article. What causes tonsil stones in my throat? The tonsil stones are the result of an excess of food particles, bacteria, mucus, and…
-
Secret Tips For Safely Getting Golden Tan Skin
Beautiful skin makes women feel more confident, and glowing tan skin is truly a dream for many. It does not only make a person look healthy and sexy, but also slimmer. However, getting a brown tan seems to require lots of time and effort, and sometimes the result is not what we wanted. Before going sunbathing, there are some precautions that should be taken. In this article, we will reveal all the secret tips for getting the perfect skin color while sharing with you useful advice from specialists. Let’s start!
-
What you should know before buying VVS diamonds
As you all know diamonds are rare and expensive. Diamonds are mostly used for beautiful rings as it is the best-known, and rarest gemstone in the world. There are many factors to consider before buying an engagement ring as a gift for someone you love. These factors vary between the color, the clarity, the cut, and the carat weight. Furthermore, a diamond’s price tag will usually give you a good idea of its quality and clarity grade. In this article, we will focus on a diamond’s clarity grade, particularly the VVS diamonds. By understanding what they are and what to expect, you will be able to make an informed decision…
-
Everything you need to know about the Black Pitbull
Humans always had a soft spot for dogs. Whether we need a friend or a protector from danger, a dog can become our best companion. Unfortunately, some people stereotype Black Pitbull dogs as dangerous. However, when it comes to outside beauty, many dog lovers prefer to pick a Black Pitbull. It is no surprise that this is one of the most sought-after Pitbull races since that beautiful and attractive fur color makes them look outstanding. You are probably wondering how much is a Black Pitbull? According to the fact that Black Pitbulls are difficult to find, you might think that they are more expensive. However, it is not always so…
-
How did the “Halloween Moon Crab” come to be known?
Do you know what a Halloween Moon Crab is? Nowadays, people want something new, and the classic home with a dog or a cat is not enough. Recently, we found out the news about an unimaginable appearance, that of a strange-looking crab. The latter presents a colorful body and also a tiny face that makes it distinct from others. In the following article, not only we are going to introduce you to the Halloween Moon Crab and its history, but we are also gonna talk about its favorite food, how to feed them, and most importantly, how to pet them. That way, you will know everything you need to know…